The FDA, SPS, and HESI are proposing a new assay called the Comprehensive in Vitro Proarrhythmia Assay (CiPA assay) to replace the clinical ICH-E14 guidelines. The FDA’s current guidelines have been successful and no drugs have been withdrawn from the market because of proarrhythmic activity since 2005. The CiVPA is intended to be a more comprehensive and efficient way to test for potential proarrhythmic activity in drugs.

Validation of intermediate risk compounds
The Comprehensive in Vitro Proarrhythmia (CiPA assay) is a model for assessing the cardioprotective risk of a wide variety of pharmaceutical and medical products. The assay is an important part of the development of new cardiac drugs and is a critical step for drug discovery and development. Although the test can be vital for the development of new cardiac drugs benefiting the heart health of many, the test has several limitations.
Validation of low risk
For this, laboratories should design a validation strategy with a minimum set of parameters that is both reproducible and low-risk. It should be based on the performance criteria of the current assay and include a calibration set. The testing process should include two steps: the training phase and the validation step. Training involves a prespecified model, which includes a set of performance criteria. In the validation step, the training drugs should be tested in the same pharmacology assays.
Validation of in silico reconstructions
The Comprehensive in vitro Proarrhythmia Assay (CiPA) is a novel safety screening proposal that is intended to replace the regulatory strategy recommended in the S7B guideline of the International Conference on Harmonization (ICH). The assay consists of three components: an in vivo assay for evaluating candidate drugs, a computational model of the human ventricular cardiomyocyte action potential, and in silico computer simulations of the cardiac system. The first component evaluates candidate drugs by assessing their effects on key cardiac ion channels. The second component uses simulations to assess whether a candidate drug can discriminate between single and multiple cardiac ion channel blockade and can accurately predict the risk associated with different types of ion channel blockade.
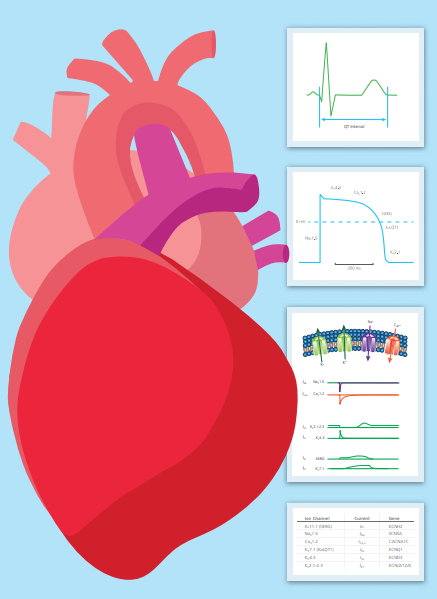
CiPA paradigm
The Comprehensive In Vitro Proarrhythmic Assay (CiPA assay) paradigm is based on a panel of in vitro and in silico assays that aim to identify the proarrhythmic risk of a wide range of drugs. In silico reconstructions of human ventricular electrophysiology were developed and patch clamp assays on human cardiac ion currents were integrated. Proarrhythmia risk was also assessed in hiPSC ventricular myocytes.
Standardization of ion channel assays
As a part of the Comprehensive in vitro Proarrhythmia Assay (CiPA) initiative, the US Food and Drug Administration (FDA) is developing guidelines to standardize and enhance cardiac safety testing. CiPA aims to improve and engineer proarrhythmic assays and create a more scientifically valid foundation for drug labelling. The CiPA consortium includes Sophion Bioscience, which has a long-standing interest in cardiac ion channels and extensive experience in cardiac safety assays.

Sophie is an early childhood education specialist with a focus on cognitive and emotional development in young children.
